-
PDF
- Split View
-
Views
-
Cite
Cite
Ewa Bielska, Robin C. May, What makes Cryptococcus gattii a pathogen?, FEMS Yeast Research, Volume 16, Issue 1, February 2016, fov106, https://doi.org/10.1093/femsyr/fov106
Close - Share Icon Share
Cryptococcosis is an invasive fungal infection of humans and other animals, typically caused by the species Cryptococcus neoformans in patients with impaired immunity. However, there is growing recognition of the importance of the related species C. gattii in causing infections in apparently immunocompetent individuals. In particular, an ongoing outbreak of cryptococcal disease in the Pacific Northwest region, which started in 1999, has driven an intense research effort into this previously neglected pathogen. Here, we discuss some of the recent discoveries in this organism from the Pacific Northwest region and highlight areas for future investigation.
INTRODUCTION
Cryptococcus gattii is a fungal pathogen of humans and other animals that can be found both as an opportunistic infection (Hagen et al.2012) and as a primary pathogen (Kwon-Chung and Varma 2006). Cryptococcus gattii is a haploid, encapsulated basidiomycete yeast that is widespread in soil, trees and tree hollows (reviewed in Springer and Chaturvedi 2010; Harris, Lockhart and Chiller 2012).
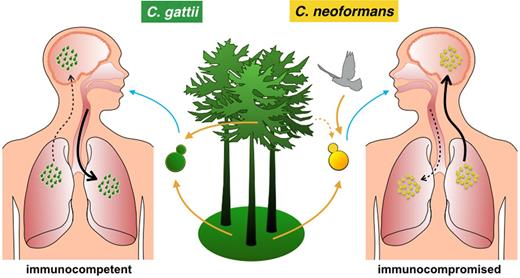
Cryptococcosis is thought to commence upon inhalation of airborne infectious propagules, such as spores or dried yeast cells, allowing the pathogen to settle in the lungs, where it can survive and proliferate within alveolar macrophages (Fig. 1). Typical symptoms that are associated with cryptococcosis are fever, weight loss, fatigue, night sweats, cough, chest pain, headache, vomiting and neck stiffness (Phillips et al.2015). If the pathogen reaches the central nervous system, this can lead to meningoencephalitis, the most severe form of cryptococcosis, which is always lethal without rapid treatment.
A schematic illustration of an infection process of C. gattii (left) and C. neoformans (right). An infection starts upon inhalation of airborne infectious propagules, which may allow the pathogen to settle in the lungs. If the fungus reaches the central nervous system, this can lead to a brain infection, which can be lethal. Note the differences between C. neoformans and C. gattii environmental origin, the immune condition of the hosts and organ preference between pathogens.
Interestingly, there is a predilection of C. neoformans for central nervous system infection and C. gattii for lung infection. On the other hand, pulmonary (and cerebral) cryptococcomas (large inflammatory masses) are formed during infection with C. gattii, but not with C. neoformans (Mitchell et al.1995; Chen et al.2000; Galanis et al.2010; Byrnes and Marr 2011); the latter leading instead mainly to small pulmonary lesions (Speed and Dunt 1995; Chen et al.2000). This might be due to higher transmigration of C. neoformans through brain blood barrier via the Trojan horse mechanism according to in vitro studies (Sorrell et al.2015).
Early reports describing patients suffering from cryptococcosis highlighted the prevalence of men over women (Chen et al.2000) which was thought due to the exposure of males to environmental sources. Recent data indicate that in fact presence of testosterone in men, but not β-estradiol in women, may influence capsule growth and reduce phagocytosis of yeast by macrophages (McClelland et al.2013; Costa et al.2015).
Interestingly, while C. neoformans mainly infects immunosuppressed patients, with HIV/AIDS being the most common underlying condition, C. gattii is considered as a primary pathogen, since it frequently infects immunocompetent and apparently healthy individuals (Speed and Dunt 1995; Sorrell 2001), although, recent studies suggest several factors such as smoking, oral corticosteroids usage and older age may increase the risk of infection by this species (reviewed in MacDougall et al.2011). Interestingly, anti-cryptococcal antibody levels are higher during C. gattii than C. neoformans infections in immunocompetent patients (Speed et al.1996) and in cats (Malik et al.1999), and thus it is possible that undiagnosed antibody deficiencies may predispose to C. gattii infections (Marr et al.2012). Similarly, high concentrations of granulocyte-macrophage colony-stimulating factor (GM-CSF) autoantibodies can be found in the plasma of otherwise healthy HIV-negative individuals suffering from cryptococcal meningitis (Rosen et al.2013) and, interestingly, are a significant risk factor for CNS infection by C. gattii but not C. neoformans (Saijo et al.2014).
In comparison to cryptococcosis caused by C. neoformans which kills 650 000 immunocompromised people suffering from HIV/AIDS every year worldwide (Park et al.2009) as well as a significant additional number of organ transplant recipients (Pappas 2013), C. gattii infections are rather rare, although recent studies indicate that they may be mis- or under-diagnosed (Iverson et al.2012; Tintelnot et al.2015). Cryptococcus gattii meningitis can be cured completely in the early stages of disease (Chen et al.2012), but often the disease is misdiagnosed as tuberculosis or other bacterial/viral pulmonary infections at this stage. Anti-fungal treatment is mainly based on amphotericin B in combination with 5-flucytosine and/or fluconazole (Chen et al.2013b) and at later stages of the disease surgery and corticosteroids may be required (Sorrell and Chen 2010).
There are many methods to differentiate between Cryptococcus species. Only C. gattii will produce blue colonies if grown on CGB (L-canavanine, glycine and bromthymol blue) agar (Kwon-Chung, Polacheck and Bennett 1982; Min and Kwon-Chung 1986). Similarly, capsular agglutination reactions can discriminate between C. neoformans, which exhibits serotypes A (C. neoformans var. grubii), D (C. neoformans var. neoformans) and AD and C. gattii (which is comprised of serotypes B and C (Kwon-Chung, Polacheck and Bennett 1982; Franzot, Salkin and Casadevall 1999; Boekhout et al.2001)). However, the most reliable methods for differentiating between Cryptococcus species are sequence based (McTaggart et al.2011; Kwon-Chung et al.2014; Hagen et al.2015) and rely on amplified fragment length polymorphism (AFLP; Boekhout et al.2001), PCR and multiplex PCR fingerprinting (Meyer et al.1999, 2003; Ito-Kuwa et al.2007) or sequencing of intergenic spacers (IGS; Diaz et al.2000, 2005).
For most of the time that cryptococcosis has been recognized, research efforts have focused on C. neoformans as the dominant pathogenic species. However, in 1999, an outbreak of cryptococcosis started on Vancouver Island (British Columbia, Canada) that was later identified as being caused by C. gattii. This outbreak subsequently spread to mainland Canada and the northwestern part of the USA (Oregon and Washington, MacDougall et al.2007). Although C. gattii was previously known to be prevalent in tropical regions (Kwon-Chung and Bennett 1984), its abrupt appearance in the moderate climate of the Pacific Northwest (PNW) region led to disease not only in otherwise healthy humans but also domestic, terrestrial and sea animals including dolphins (Stephen et al.2002; Kidd et al.2004; MacDougall et al.2007; Upton et al.2007). During 1999–2007, the outbreak affected 218 people (5.8 people per million in the region per year) and 19 died (8.7% associated deaths; Galanis et al.2010; Phillips et al.2015). In addition, during 2004—2011, the outbreak affected approximately 100 people, with a 33% mortality rate, in the US PNW (Harris et al.2011). Most people suffered from respiratory illness (76.6%) or lung cryptococcomas (75.4%; Galanis et al.2010), with a third of those patients also showing central nervous system infection (Phillips et al.2015).
97% of documented cryptococcosis cases from the Vancouver Island outbreak were compromised of molecular genotype VGII (VG = variety gattii; Boekhout et al.2001; Kidd et al.2004), with subtype VGIIa being responsible for 86.3% of cases in BC (Galanis et al.2010) and 81% in Washington/Oregon (Harris et al.2011). The remaining cases were caused by a closely related lineage, VGIIb (also termed the minor lineage; Kidd et al.2004), while a third lineage, VGIIc (novel) has been reported mainly from Oregon State (Harris et al.2011). VGIIa exhibits higher fertility than other C. gattii strains (Ngamskulrungroj et al.2008) and the molecular type is now considered as the most virulent within this species (Lizarazo et al.2014).
In comparison to non-outbreak isolates from Australia, where C. gattii is endemic, the majority of the Vancouver Island outbreak isolates were highly fertile (70% in BC versus 10% in Australia; Fraser et al.2003), hypervirulent (Fraser et al.2005) and showed low susceptibility to anti-fungal drugs (Trilles et al.2012). Although detailed analysis of many C. gattii isolates has been performed, no individual cryptococcal pathogenicity factors have yet been found (Ma et al.2009), leading to the suggestion that virulence is a complex, multifactorial phenotype (Garcia-Solache et al.2013; Firacative, Duan and Meyer 2014). Below we discuss some of the features of this pathogen which may contribute to this multifactorial pathogenicity.
ENVIRONMENTAL NICHE OF C. GATTII
While C. neoformans spores can be found in birds’ droppings, the environmental presence of C. gattii is strictly associated with plants. To date, 54 species of trees growing globally (Hagen and Boekhout 2010; Springer and Chaturvedi 2010) have been found to host C. gattii, with Australian eucalyptus (Ellis and Pfeiffer 1990), almond trees (Callejas et al.1998) in tropical and semi-tropical regions and, quite recently, Pseudotsuga menziesii (Oregon pine) in regions with a more moderate climate (Springer and Chaturvedi 2010) acting as dominant host species. Cryptococcus gattii can proliferate and mate on plant surfaces rich in myo-inositol (Xue et al.2007; Springer et al.2010) and there is some evidence that C. gattii may persist longer in the environment in the presence of plant tissue (Huerfano, Castaneda and Castaneda 2001).
THE ORIGIN OF THE PNW OUTBREAK
Early investigations proposed several sources for the VGII outbreak strain, but more recent work points to an origin in South America (VGIIa; Hagen et al.2013; Billmyre et al.2014; Engelthaler et al.2014) and Australia (VGIIb; (Fraser et al.2005; Billmyre et al.2014)). Separation of the VGI and VGII C. gattii strains occurred around 12.4 million years ago (D'Souza et al.2011), and this extended period of genetic isolation has contributed to recent suggestions to raise these lineages to species level (Hagen et al.2015). Although the molecular genotype of the VGII population differs significantly from other strains of C. gattii, due to mutations and recombinations, within the VGII group, the four subpopulations VGIIa, VGIIb, VGIIc and VGIIx are highly clonal and not very diverse (Billmyre et al.2014; Engelthaler et al.2014; Farrer et al.2015). A comparison between genomes from VGIIa isolates suggests that these groups diverged less than 100 years ago from a less virulent strain in which a frameshift mutation in a DNA repair gene msh2 was found (Billmyre et al.2014). VGIIa genomes from the outbreak do not contain the frameshift mutation, and it has been hypothesized that its genome reverted via mitotic microevolution (Billmyre et al.2014). If this is true, there is a possibility that the reversion occurred after gaining adaptation for higher virulence. Since the other non-pathogenic VGIIa-like isolates have retained this hypermutator mutation, it remains possible that such an event may recur leading to the emergence of novel outbreak strains (Billmyre et al.2014).
In addition, several studies have highlighted the potential for gene transfer (introgression) occurring between C. neoformans var. grubii and C. gattii (Engelthaler et al.2014) as well as between different C. gattii clades (Billmyre et al.2014) through sexual reproduction (bisexual or unisexual). Support for the latter model comes from evidence for genomic islands of high polymorphism within a VGIIa genome, which were potentially introduced from two distinct VGII clades (Billmyre et al.2014).
To date, all clonal C. gattii VGII isolates identified from the PNW have been mating type MATα (Lockhart et al.2013). Unlike MATa strains, MATα strains are capable of same-sex mating (Wiesner et al.2012) and this has been proposed as a potential origin for the PNW outbreak (Fraser et al.2005). In C. neoformans, MATα strains are also associated with higher virulence (Kwon-Chung, Edman and Wickes 1992; Barchiesi et al.2005; Nielsen et al.2005), but this appears not to be the case with C. gattii (Zhu et al.2013).
GENOMIC DIFFERENCES
Separation between the VGI and VGII C. gattii strains has resulted in significant genome differences including chromosomal rearrangements and higher than expected overall nucleotide sequence divergence (D'Souza et al.2011). Recently, a whole genome analysis was performed for all lineages of C. gattii (Farrer et al.2015), where the authors compared nuclear and mitochondrial DNAs between lineages. Interestingly, they found that the PNW outbreak VGII lost 146 genes (three times more than number of genes lost in VGI–III–IV lineages combined) that are still present in the three other lineages, including a mitochondrial cytochrome c peroxidase gene and several other genes that are typically thought of as being essential for nuclear and mitochondrial genome maintenance. At the same time, the VGII lineage has gained several unique genes encoding proteins with COX6B, HSP70 and iron-binding domains and proteins possibly involved in membrane trafficking.
LACK OF RNA INTERFERENCE MACHINERY
One of the most remarkable discoveries to emerge from the extensive genome sequencing effort in this species is the VGII-specific absence of genes encoding Argonaute, Ago1 and Ago2 (D'Souza et al.2011), which are critical components of the RNA interference (RNAi) machinery in other fungi including C. neoformans (Janbon et al.2010). This lack of ago genes was found in all VGII isolates, including those from beyond the PNW region (Farrer et al.2015). Thus, the C. gattii VGII lineage is lacking an RNAi-mediated genome defense during both the sexual cycle (Wang et al.2010; Dumesic et al.2013) and vegetative growth (Wang et al.2012). This loss of silencing machinery appears to have independently occurred in several pathogens (reviewed in Nicolas, Torres-Martinez and Ruiz-Vazquez 2013), a finding which remains enigmatic. However, this loss of RNAi typically leads to transposon reactivation, which may accelerate genome evolution and potentially help in developing novel anti-host mechanisms (Oliver and Greene 2009; Biemont 2010; Wang et al.2010). Alternatively, the loss of RNAi may be a protective response to pathogens that can otherwise hijack this pathway. Cross-kingdom hijacking of RNAi silencing is known for other pathogens, such as the plant fungal pathogen Botrytis cinerea which is able to hijack Arabidopsis and tomato RNAi machineries by binding to host AGO1, leading to the silencing of host immunity genes and facilitating infection (Weiberg et al.2013). To date, pathogens of Cryptococci have not been identified, but such a hypothesis remains at least a theoretical possibility.
FERTILITY
Although Cryptococci can reproduce sexually, where two opposite mating types, MATa and MATα, mate and produce spores, a predominance of α mating type in the environment means that alternative reproduction strategies are common. In particular, same-sex mating between two α mating-type parents (Lin, Hull and Heitman 2005), or spontaneous generation of spores by haploid strains (known as monokaryotic fruiting) can both produce infectious propagules. It has been suggested that the rapid expansion of the PNW outbreak has been driven primarily by clonal reproduction (Fraser et al.2005) and it is therefore enigmatic that the majority of the PNW outbreak isolates are highly fertile (Fraser et al.2003; Ngamskulrungroj et al.2008). Despite this, whole genome analysis has revealed very limited nuclear genetic exchange between C. gattii lineages (Farrer et al.2015), although interestingly several instances of recombination within the mitochondrial genome (Voelz et al.2013; Farrer et al.2015).
INFLAMMATION AND THE CYTOKINE RESPONSE
In contrast to C. neoformans, C. gattii is able to infect immunocompetent individuals, suggesting that the latter uses different or additional methods to inhibit immune responses. Somewhat counterintuitively, in human peripheral blood mononuclear cells, C. gattii induces higher concentrations of cytokines such as pro-inflammatory interleukin IL-1β, TNF- α and IL-6 and the T-cell cytokines IL-17 and IL-22 than C. neoformans (Schoffelen et al.2013). Interestingly, however, the authors found that Toll-like receptor (TLR) 4 and TLR9 were involved in the recognition of the pathogen, but not TLR2, unlike C. neoformans (Vecchiarelli 2005). These results suggested that a different innate cytokine response of the host might be related to different pathogen-activated molecular pattern (PAMPS) molecules localized on the C. gattii surface in comparison to C. neoformans. TLR2 is known to recognize chitin (Da Silva et al.2008). Chitin-like structures in Cryptococci are only exposed in the limited parts of the cell surface under the capsule (Rodrigues et al.2008b), which may be the reason why they are not normally recognized by TLR2. Thus it is possible that differences in the organization and localization of chitin-derived structures between C. neoformans and PNW C. gattii strains might explain different preferences in organ colonization, since C. gattii preferentially targets the lungs, whilst the brain is the primary target organ for C. neoformans (Ngamskulrungroj et al.2012; Sorrell et al.2015).
Although in vitro blood infections by C. gattii result in potent inflammatory signaling, in pulmonary tissue Hoang et al. found only minimal inflammatory responses to C. gattii (Hoang et al.2004). This may be accounted for by the ability of C. gattii to weaken pulmonary Th1 and Th17 responses (at least in mice) via altered dendritic cell (DC) function through down-regulation of pulmonary chemokine expression (Angkasekwinai et al.2014). This restricted DC function is related to reduced levels of TNF-α, and indeed addition of recombinant TNF-α fully restores DC maturation and thus T cell responses (Huston et al.2013).
Thus acute introduction of C. gattii may induce rapid inflammation, but longer lasting systemic inflammation is dampened by poor DC activation. This biphasic response may also explain otherwise contradictory findings, such as the relatively slower growth of C. gattii than C. neoformans in blood (Ngamskulrungroj et al.2012) (suggesting strong induction of defense) and yet reduced neutrophil infiltration to sites of infection (Cheng, Sham and Kronstad 2009).
VIRULENCE STRATEGIES
Human fungal pathogens often use a huge repertoire of virulence strategies in order to survive inside the host. In cryptococci, the polysaccharide capsule, chitin and melanin within the cell wall, phospholipases, urease, laccase and the ability to growth at 37°C are the most studied virulence factors involved in pathogenesis. However, although these features are shared by all pathogenic Cryptococci, there are some crucial differences among them that might play a role in the hypervirulence of PNW C. gattii isolates.
GROWTH AT 37°C
The ability to survive at elevated temperature is crucial for human pathogens. In Cryptococci, this is regulated by Calcineurin, a Ca2+/calmodulin-activated serine/threonine-specific phosphatase (Liu et al.1991). Typically, mutants lacking calcineurin gene, such as cna1Δ, are avirulent in both C. neoformans and C. gattii isolates. However, PNW VGIIa strains lacking calcineurin function are still viable at elevated temperature (Chen et al.2013a), suggesting there may be as-yet unidentified differences in temperature tolerance in this lineage.
CAPSULE
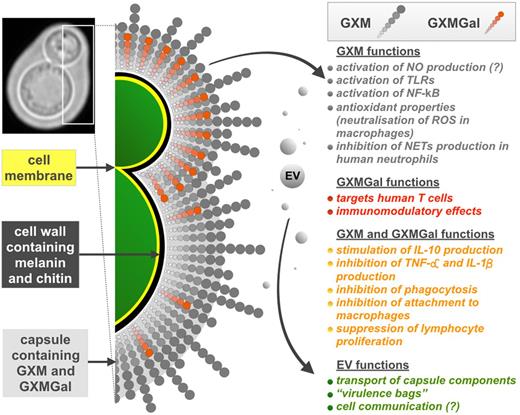
Cryptococcal capsule is a highly hydrated and a negatively charged mesh of polysaccharides surrounding the yeast cell (Fig. 2), mainly composed of glucuronoxylomannan (GXM; composed of mannose, xylose and glucuronic acid), and glucuronoxylomannogalactan (GXMGal) plus mannoproteins (Vartivarian et al.1989), and its growth is activated during host infection. Most studies to date have focused on C. neoformans capsule, which is considered a major virulence factor (McClelland, Bernhardt and Casadevall 2006) and has anti-phagocytic properties in macrophages (Kozel and Mastroianni 1976). This is correlated with a reduction of systemic inflammation (reviewed in Vecchiarelli et al.2013) mainly due to a suppression of T-lymphocyte proliferation (Syme et al.1999), induced secretion of the anti-inflammatory cytokine IL-10 (Vecchiarelli et al.1996) and inhibited secretion of TNF-α and IL-1β (Vecchiarelli et al.1995) by human monocytes.
Diagram representing the role of cryptococcal polysaccharide capsule and its involvement in several immune responses. Chitin-like structures (not shown) composed of β-1,4-N-acetylglucosamine oligomers link the capsule to the cell wall (Rodrigues et al.2008a). GXMGal molecules (shown in red) are mainly found in growing capsules of budding daughter cell (De Jesus et al.2009) and also in the capsules of mature cells but only transiently due to secretion (De Jesus et al.2010).
GXM is a large molecule (around 4600 000 Daltons in serotype B strain I23) and has different structures (McFadden, De Jesus and Casadevall 2006) which correlate with differences in antibody reactivity (Fonseca et al.2010). Capsule size depends on environmental conditions (summarized in Zaragoza and Casadevall 2004; Gupta and Fries 2010) and capsule enlargement is usually observed during infection (Garcia-Hermoso, Dromer and Janbon 2004). This is linked to the presence of mammalian serum (Zaragoza et al.2003), higher CO2 concentration (Granger, Perfect and Durack 1985) and tissue-specific conditions such as iron deficiency in the lungs (Vartivarian et al.1993; Rivera et al.1998) or the high concentration of urea in cerebrospinal fluid (Frazzitta et al.2013). In addition, capsular size can change during the cell cycle and its enlargement is mainly observed during the G1 phase when no budding occurs (Garcia-Rodas et al.2014).
Depending on the C. gattii strain, age of the cells, temperature, conditions and methodology used for studies, capsule thickness can differ dramatically. For instance, relative to the canonical C. neoformans strain H99 (McFadden, De Jesus and Casadevall 2006), C. gattii capsules can be very similar (NIH191 and NIH198; Frases et al.2009), much smaller (strains CN23/10.993 and the PNW strain R265 (Cheng, Sham and Kronstad 2009; Fonseca et al.2010)) or significantly larger (strain I23 and R265; Frazzitta et al.2013).
Although the major capsular polysaccharide GXM is generally immunosuppressive, fractions with molecular masses below 10 000 Daltons isolated from C. gattii strains were effective in stimulating nitric oxide (NO) production by host macrophages and in activation of TLRs (TLR2/1 and TLR2/6) and NF-κB (Fonseca et al.2010). Increased production of NO has also been observed after incorporation of extracellular vesicles (EVs) by murine macrophages, but it was diminished after adding fractions of GXM (Oliveira et al.2010), suggesting that these two components may act in concert to reduce host inflammatory responses.
Interestingly, the cryptococcal capsule is also likely to play an important role in the environment. On plant surfaces, some strains of C. gattii form 40–100 nm length extracellular fibrils which then allow yeast cells to escape from human neutrophils in vivo, potentially by inhibiting the production of neutrophil extracellular traps (Rocha et al.2015). Consequently, infection of mice with yeast cells grown on leaf agar was more severe and showed higher proliferation in the lung and brain then when yeast cells grown on YPD agar were used (Springer et al.2010).
EXTRACELLULAR VESICLES
GXM, the main polysaccharide component of the cryptococcal capsule, is synthesized intracellularly and transferred from the Golgi apparatus (Hu et al.2007) to the outside of the cell wall via EVs (Yoneda and Doering 2006). The bilayered membrane-EVs serve not only as transporting ports for capsule components, but also are used by cryptococci as 20–400 nm diameter ‘virulence bags’ (Rodrigues et al.2007, 2008a). Studies performed so far on C. neoformans revealed that EVs contain ribosomal proteins as well as proteins related to virulence and anti-oxidant defense, including laccase (melanin synthesis), urease, superoxide dismutase and heat shock proteins (Rodrigues et al.2008a; and reviewed in Rodrigues et al.2014). Interestingly, a C. neoformans sec 6 RNAi mutant, which is impaired in EV secretion, was attenuated in virulence in mice, although growth at 37°C, capsule formation and phospholipase activity were not affected (Panepinto et al.2009). Recent studies on EVs from different fungi including C. neoformans revealed that these vesicles are packed with a spectrum of short non-coding mRNAs, which are thought to play a role in cell communication and pathogenesis (Peres da Silva et al.2015). Unfortunately, there is no data regarding function and content of EVs isolated from C. gattii to date.
On the other hand, cryptococcal EVs can enhance host anti-microbial activity after incorporation by murine macrophages where increased levels of NO and cytokines (extracellular TNF-α, IL-10 and transforming growth factor TGF-β) were observed (Oliveira et al.2010). Similar results were obtained after treatment of macrophages with EVs isolated from Candida albicans (Vargas et al.2015) suggesting that EVs can serve as a platform of secreted virulence for pathogenic fungi.
At first glance, enhancing host phagocytosis in this way seems like a disadvantageous step for a pathogen. However, since C. gattii can happily survive within the phagosome and, at the same time, be protected from other immune cells as well as extracellular anti-fungal molecules such as complement, it may be that EV-induced boosting of phagocytosis offers survival advantages to pathogens such as C. neoformans and C. albicans by facilitating their entry into an intracellular niche (Oliveira et al.2010; Vargas et al.2015).
SURVIVAL WITHIN MACROPHAGES
Although Cryptococci are phagocytosed by macrophages, in most cases they can then survive and proliferate inside these host cells. Cryptococci have developed an amazing repertoire of anti-phagocytic strategies (reviewed in Johnston and May 2013), most probably as a result of prolonged selective pressure from environmental predators such as amoebae (Steenbergen, Shuman and Casadevall 2001). As a result, virulence and defense mechanisms against phagocytic cells could be acquired and selected during the evolution of fungus–amoebal interactions in the environment. In support of this model, transcriptional profiles show strong similarities between genes upregulated by yeast internalized by amoebae and murine macrophages (Derengowski Lda et al.2013). Interestingly, however, relative to C. neoformans, C. gattii is rarely phagocytosed by the model amoeba Acanthamoeba castellanii, perhaps reflecting differences in their capsule structure (Malliaris, Steenbergen and Casadevall 2004).
FUNGAL PERSISTENCE AND REACTIVATION
Cryptococcus neoformans is classically thought of as a long-term latent pathogen that reactivates upon immunocompromisation, but this picture is more complex for C. gattii. Recent multilocus sequence typing between European and worldwide isolates has revealed that dormant C. gattii infections can be reactivated many years after the initial infection (Hagen et al.2012), for instance following treatment with corticosteroids (Hagen et al.2010). However, unlike C. neoformans, it appears that many C. gattii infections represent de novo acquisition of the organism from the environment, rather than (re)activation of latent disease (MacDougall and Fyfe 2006).
PROLIFERATION INSIDE MACROPHAGES AND ‘DIVISION OF LABOR’
All pathogenic Cryptococci appear capable of survival and proliferation within macrophages. However, outbreak C. gattii isolates are capable of intracellular proliferation rates that exceed those of all other isolates and which correlate with virulence (Ma et al.2009). In contrast, C. neoformans virulence is associated to macrophage uptake and laccase activity, but not to intracellular proliferation rate (IPR; Sabiiti et al.2014). This difference offers a potential explanation for their varying host profiles; C. gattii infections in otherwise healthy individuals can only proceed if intracellular proliferation is rapid enough to overwhelm the host immune system. In contrast, C. neoformans infections in immunocompromised hosts instead rely on ‘stealth’, in which rapid proliferation is not necessarily beneficial but an intracellular niche is critical for survival.
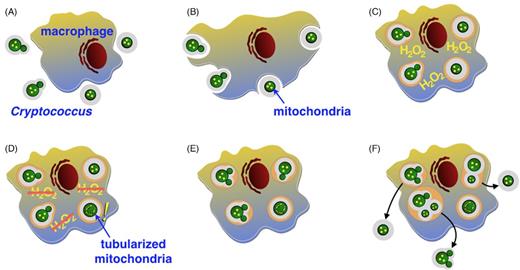
In the case of C. gattii outbreak isolates, rapid intracellular proliferation is associated with changes in mitochondrial morphology (Ma et al.2009; Voelz et al.2014). It was initially proposed that this change in mitochondrial morphology could protect the pathogen against the intracellular environment of the phagocytic cells (Ma et al.2009; Ma and May 2010). However, more recent analyses of this group have indicated a more complex and intriguing model. Upon entry into host phagocytes PNW outbreak strains of C. gattii trigger a ‘division of labor’ mechanism in which some cells adopt this mitochondrial morphology and cease division, but in doing so they facilitate extremely rapid proliferation of neighboring Cryptococci, thus driving amplification of the population as a whole (Fig. 3 ; Voelz et al.2014).
Diagram representing a scheme of ‘division of labor’ during engulfment of cryptococcal cell by a macrophage. (A) Receptor-mediated phagocytosis allows recognition of the fungal cell. (B) Phagocytosis of the pathogen cell by an alveolar macrophage. (C) One of the first steps of host defense is an oxidative burst during when macrophage releases reactive oxygen species. (D) A subpopulation of guardian cells sacrifice their proliferation and tubularize their mitochondria which is accompanied with reduction of host ROS (E) This allows proliferation of neighboring cryptococcal cells (F) and following escape from the macrophage.
Surprisingly, a comparison of mitochondrial genomes between C. gattii and C. neoformans revealed similar gene content (D'Souza et al.2011). Likewise, a very recent whole genome analysis did not identify any single gene that is characteristic of the PNW strains (Farrer et al.2015). However, several studies have highlighted unusual patterns of mitochondrial inheritance and recombination in this lineage (Bovers et al.2009; Xu, Yan and Guo 2009; Voelz et al.2013), suggesting that unusual combinations of nuclear and mitochondrial alleles may contribute to the virulence of this group.
Interestingly, the inheritance patterns of mitochondria in C. gattii can be influenced by several environmental variables including UV exposure and higher temperatures (Wang, Wilson and Xu 2015), suggesting an intriguing link between environmental conditions and the evolution of novel genotypes in this group.
ESCAPING
In addition to intracellular proliferation, Cryptococci can also escape from host cells in a poorly understood process called vomocytosis (Alvarez and Casadevall 2006; Ma et al.2006). Interestingly, the frequency of this non-lytic expulsion process in vivo seems to be higher than the rates obtained in vitro (Nicola et al.2011). There is considerable interest in the contribution that vomocytosis may make to tissue dissemination by allowing infected phagocytes to ‘deposit’ Cryptococci at distant sites; the so-called Trojan Horse model. Charlier and colleagues have previously provided evidence for this mechanism of entry across the blood–brain barrier (Charlier et al.2009), although recent work using C. neoformans mutants with reduced phagocytosis by macrophages showed no difference in rates of CNS entry (Tseng et al.2012). Both C. neoformans and C. gattii undergo vomocytosis in vitro, and rates appear similar, at least between C. neoformans H99 and C. gattii R265 (Voelz, Lammas and May 2009) suggesting that differential vomocytosis is unlikely to be a major factor in PNW virulence. Rather, it appears that the slower growth of PNW C. gattii isolates in blood (10–100 times slower than C. neoformans) coupled with their exceptionally fast replication within host cells means that C. gattii infections frequently present as pulmonary infections rather than disseminated CNS disease (Ngamskulrungroj et al.2012; Sorrell et al.2015).
CELL GIGANTISM
Enlargement of the cryptococcal capsule has been documented as a mechanism of protection against phagocytosis and the phagocytic oxidative burst for many years (Zaragoza et al.2008). However, recently an additional role for cell size increase has become apparent. During in vivo infections, giant or ‘titan’ cells (50–100 μm in diameter) form and represent about 20% of the cryptococcal population during pulmonary infection (Okagaki et al.2010; Zaragoza et al.2010). Intriguingly, the presence of titan cells in the cryptococcal population reduces overall phagocytosis (not just of the titan cells themselves) by macrophages (Okagaki and Nielsen 2012). Recent studies using the moth larvae G. mellonella showed that during C. gattii infections, both the capsule and the cell sizes of VGII cells underwent significant enlargements up to 75 μm, but this was not observed in a very virulent PNW isolate R265 (Firacative, Duan and Meyer 2014). These observations are consistent with a suggestion that cell ‘titanisation’ provides an additional defense mechanism of the isolates attenuated in virulence (Evans et al.2015) and/or that this strategy is critical for long-term latent infections, but perhaps less vital for highly virulent acute infections caused by PNW strains.
CONCLUSIONS
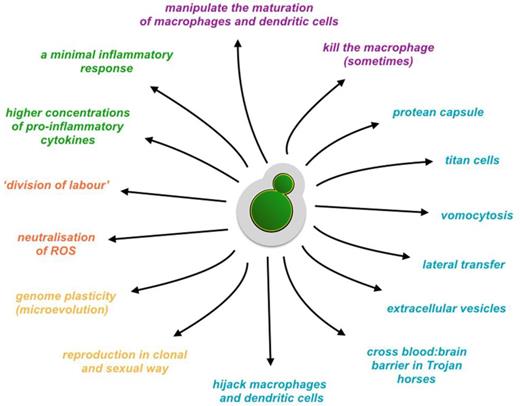
Recent epidemiological data indicate that the PNW outbreak of C. gattii infection is receding, although the fungus now appears endemic to Vancouver Island (Espinel-Ingroff and Kidd 2015; Kwon-Chung and Saijo 2015), However, understanding the apparently recent and dramatic evolutionary history of virulent VGIIa isolates is of profound importance both for improving our understanding of fungal pathogenesis (Fig. 4) in general and for determining the likelihood of other such outbreaks in the near future.
A cartoon representing a repertoire of cryptococcal pathogenic activities.
FUNDING
The authors are supported by funding from the European Research Council Award ‘MitoFun’ (RCM & EB) and by a Lister Institute Fellowship and a Royal Society Wolfson Merit Award (RCM).
Conflict of interest. None declared.
REFERENCES